

Best pH Sensor for Bioreactors
Maintaining the correct pH inside a bioreactor is essential for successful fermentation, cell culture, and bioprocess optimization. Even small pH fluctuations can dramatically affect enzyme
# Type at least 1 character to search # Hit enter to search or ESC to close

No products in the cart.

No products in the cart.
Product Categories

Cleaning a pH probe properly involves more than just maintenance. Thorough cleaning helps ensure accurate measurements, prolongs the life of the probe, and fulfills compliance requirements.
When it comes to any introductory course in analytical chemistry and process monitoring, few people tend to misunderstand and overlook the importance of a pH probe.
Atlas Scientific’s laboratory-grade pH probes, industrial ORP sensors, and precision conductivity probes are just a few examples of electrochemical sensors that greatly aid numerous laboratories, manufacturing sites, and environmental monitoring stations all over the globe. In the last few decades, the frequency of pH probe usage has soared, while at the same time, the maintenance tends to be the most neglected part of the analytical measurements.
Overlooking the importance of a pH probe can result in disastrous production mistakes, failed quality control tests, and compliance with regulations, compromising the data collected from thorough research. Take the example of pharmaceutical manufacturing: A single pH reading error can invalidate a production batch, costing way over a million dollars.
In this guide, we will transform your understanding of pH probe cleaning that will maximize accuracy, extend probe life, and ensure you receive consistent and reliable measurements across your applications.
pH probe contamination involves far more than visible deposits, encompassing a complex interplay of chemical, biological, and physical fouling mechanisms that can dramatically compromise sensor performance.
Protein fouling presents particularly challenging problems in biological applications, where proteins denature and precipitate onto glass membranes, creating insulating barriers that slow response times and reduce sensitivity. This contamination proves especially problematic in fermentation monitoring, food processing, and clinical applications where protein concentrations run high.
Inorganic precipitation occurs when metal oxides, sulfides, and other inorganic compounds form crystalline deposits directly on probe surfaces. These deposits appear frequently in industrial wastewater treatment, mining operations, and geothermal applications where dissolved mineral concentrations reach extreme levels.

Organic film formation develops when oils, surfactants, and similar compounds create hydrophobic films that prevent proper electrode hydration. This contamination type frequently appears in petrochemical processes, food manufacturing, and agricultural applications where organic compounds are prevalent.
Ion-specific interference emerges when certain ions selectively bind to glass membranes, fundamentally altering electrochemical properties and creating systematic measurement errors. Heavy metals like lead and mercury prove particularly problematic in this regard, requiring specialized cleaning approaches.
Microbial biofilms represent perhaps the most challenging form of pH probe contamination, creating complex matrices of bacteria, fungi, and extracellular polymeric substances that form living barriers continuously evolving and adapting to resist cleaning attempts.

Biofilms create particularly difficult challenges because they:
Effective pH probe cleaning requires understanding specific contamination mechanisms and selecting appropriate cleaning strategies rather than simply applying the strongest available chemicals. This systematic approach must be grounded in contamination analysis, chemical compatibility assessment, and fundamental electrochemical principles.

Before selecting any cleaning method, conduct thorough contamination assessments using these essential steps:
We recommend using 0.4% HCl with 5g/L pepsin enzyme, a superior formulation compared to commonly cited 0.1M HCl solutions. This enhanced approach requires 2-4 hours contact time for heavy protein contamination, significantly longer than typical 30-60 minute protocols, with optimal temperature maintained at 37°C for maximum enzyme activity.
The enhanced procedure begins with rinsing probes using deionized water to remove loose debris, followed by immersion in pre-warmed cleaning solution, ensuring complete submersion of Atlas Scientific’s glass membrane and reference junction. For Atlas Scientific probes with integrated temperature compensation, ensure temperature sensors remain fully submerged throughout the process. Gently agitate solutions every 30 minutes to enhance mass transfer while monitoring cleaning progress through impedance measurements when available.

Complete the process with thorough deionized water rinsing using a minimum of three rinse cycles, neutralization in a pH 7 buffer for 15 minutes, and full recalibration using fresh buffer solutions.
This protocol works superior to standard methods because the higher acid concentration (0.4% versus 0.1%) combined with extended contact time, provides superior protein removal compared to conventional protocols. This proves particularly important for Atlas Scientific’s precision glass membranes, which require complete protein removal to maintain fast response times.
We recommend 4% HCl for basic process fouling or 4% NaOH for acidic process fouling, with contact times of 5-10 minutes at room temperature. Begin by identifying contamination sources, using acid for basic processes or base for acidic processes. For Atlas Scientific ORP sensors, ensure platinum bands remain fully submerged throughout cleaning.
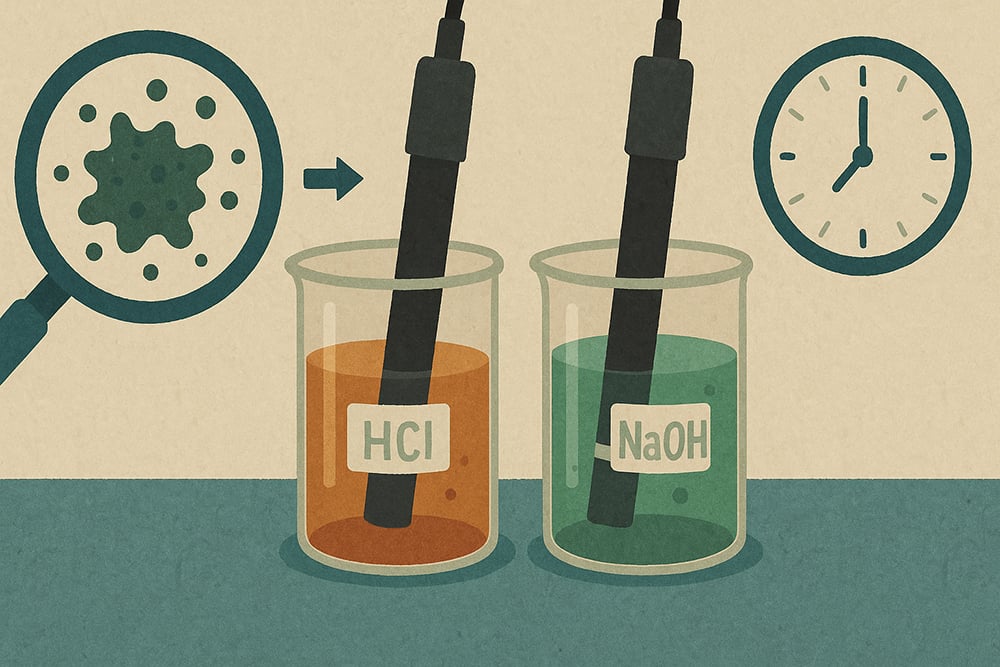
Immerse probes in cleaning solutions with gentle agitation, remembering the critical safety requirement to never alternate between acid and base without thorough intermediate rinsing. Complete with thorough potable water rinsing using a minimum of five rinse cycles, conditioning in 3M KCl storage solution for 10 minutes, and full calibration verification.
This approach beats standard methods because the 4% concentration provides dramatically faster cleaning times compared to typical 0.1M solutions while remaining safe for Atlas Scientific’s robust probe construction, saving significant downtime in production environments.
Use 5% sodium hypochlorite solution with pH adjusted to 11-12, requiring 2-4 hours contact time at room temperature, though stubborn biofilms may require overnight treatment.
Pre-rinse with sterile deionized water before immersing probes in alkaline hypochlorite solution, ensuring complete submersion of glass membranes and reference junctions.
Periodically check cleaning progress while maintaining safety protocols. Neutralize residual chlorine with sodium thiosulfate solution, rinse extensively with sterile water, condition in sterile pH 4 buffer for 30 minutes, and recalibrate with sterile buffer solutions.
Always work in well-ventilated areas when using hypochlorite solutions, and never mix with acidic solutions due to toxic chlorine gas formation.

Use 50% isopropanol in deionized water with 1% surfactant for 15-30 minutes at room temperature. Gently wipe external contamination with lint-free cloths before immersing probes in cleaning solutions.
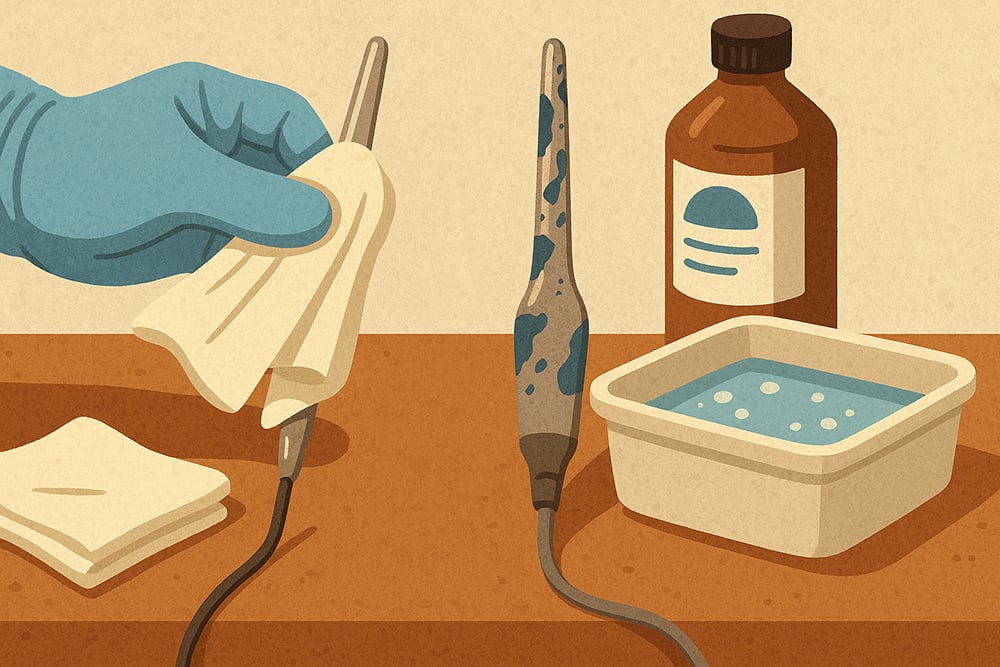
Agitate gently to enhance solubilization, rinse thoroughly with deionized water, and check for complete removal of hydrophobic films. Complete with conditioning in a pH 4 buffer solution and calibration accuracy verification.
For probes with particularly stubborn contamination, ultrasonic cleaning provides enhanced efficacy without mechanical damage.
Use low-frequency (40 kHz) ultrasonic baths with appropriate cleaning solutions for 5-10 minute intervals, maintaining power density at 0.5-1.0 W/cm³, temperatures below 40°C to prevent probe damage, and ensuring cleaning solutions are properly degassed before use.

Advanced enzymatic cleaning systems use specific enzymes targeted to particular contamination types, including proteases for protein contamination, lipases for fat and oil deposits, cellulases for plant-based organic matter, and amylases for starch-based contamination. These systems often provide superior cleaning efficacy while being gentler on probe materials than harsh chemical cleaners.
Water quality proves fundamental, always requiring Type II or better deionized water (resistivity >1 MΩ·cm) for cleaning solution preparation, since trace contaminants in lower-quality water can interfere with cleaning efficacy and introduce measurement errors.
Use analytical-grade reagents whenever possible, as industrial-grade chemicals may contain impurities that contaminate probes or interfere with subsequent measurements. Prepare fresh cleaning solutions regularly, with most solutions requiring use within 24-48 hours of preparation to maintain optimal cleaning efficacy.
Implement regular quality control checks, including pH verification using calibrated meters, conductivity testing to monitor ionic strength consistency, visual inspection for precipitation, discoloration, or contamination, and efficacy testing through periodic validation using controlled contamination studies.

Effective pH probe cleaning requires robust documentation and standardized procedures. Cleaning logs should include date and time of cleaning, contamination type and severity assessment, cleaning protocols used, contact times and temperatures, pre- and post-cleaning calibration data, and technician identification and training status.
Procedure standardization must encompass detailed step-by-step protocols, safety requirements, and personal protective equipment (PPE) specifications, quality control checkpoints, troubleshooting guidelines, and comprehensive documentation requirements.
Develop comprehensive training programs covering contamination identification techniques, proper cleaning protocol selection, safety procedures and chemical handling, documentation requirements, and troubleshooting common issues. Regular competency assessments ensure consistent cleaning quality across all personnel.
Incomplete protein removal manifests through slow response times, calibration drift, and visible protein deposits, typically caused by insufficient enzyme activity, inadequate contact time, or suboptimal temperature. Solutions include increasing enzyme concentration or contact time, verifying enzyme storage and activity, optimizing temperature to 37°C for maximum enzyme activity, and considering sequential cleaning with different enzyme types.

Persistent biofilm formation appears as recurring biological contamination and erratic readings in sterile applications, usually resulting from incomplete biofilm removal, inadequate disinfection, or contaminated storage solutions. Address these issues by extending hypochlorite contact time, increasing solution pH for enhanced biocidal activity, implementing regular preventive cleaning schedules, and using sterile storage solutions between measurements.
Chemical residue interference presents as calibration errors, baseline drift, and unusual response characteristics, typically caused by insufficient rinsing, incompatible cleaning chemicals, or residual cleaning solutions. Solutions involve extending rinse procedures with multiple water changes, implementing intermediate neutralization steps, using cleaning solution compatibility matrices, and performing cleaning solution blank measurements.
Develop cleaning schedules based on application-specific contamination risk levels:
Proper storage requires a pH 4 buffer solution with antimicrobial preservative for standard storage, saturated KCl solution for reference electrode stability during long-term storage, and dry storage only for specific probe types following manufacturer specifications.
Storage environments should maintain temperatures between 4-25°C while avoiding freezing, provide light protection through dark storage conditions, prevent contamination using sealed storage containers, and require monthly solution replacement.

Proper pH probe cleaning delivers significant economic benefits through direct cost savings, including extended probe lifespan improvements of 2-3 times typical performance, reduced calibration frequency and reagent consumption, decreased downtime for maintenance and troubleshooting, and lower replacement probe inventory requirements.
Indirect benefits include improved measurement accuracy and precision, reduced risk of batch failures and product recalls, enhanced regulatory compliance, and improved process optimization and efficiency. ROI calculations show that typical industrial pH probes costing $200-800 can benefit from cleaning protocols costing $10-20 per cycle, with extended probe life from 6 months to 18 months, providing an ROI of 300-500%.
Proper pH probe cleaning represents far more than routine maintenance, serving as a critical analytical skill that directly impacts measurement quality, operational efficiency, and economic performance. By understanding contamination mechanisms, implementing systematic cleaning protocols, and maintaining rigorous documentation practices, analytical professionals ensure their pH measurements meet the highest standards of accuracy and reliability.

The investment in proper cleaning procedures, measured in time, training, or resources, invariably pays dividends through extended probe life, improved measurement quality, and reduced operational costs. As analytical requirements become increasingly stringent and applications more demanding, mastery of pH probe cleaning remains an essential competency for analytical professionals across all industries.
If you require additional advice with cleaning probes or would like to learn more about our probes and calibration solutions, do not hesitate to contact the world-class team at Atlas Scientific.

Maintaining the correct pH inside a bioreactor is essential for successful fermentation, cell culture, and bioprocess optimization. Even small pH fluctuations can dramatically affect enzyme

Reverse osmosis water is best for hydroponics, providing near-zero EC and removing contaminants. It offers reliable results, precise nutrient control, and typically pays for itself
Notifications