

Understanding COD in Wastewater Treatment
The amount of oxygen needed to chemically oxidize organic and inorganic materials in wastewater is measured by Chemical Oxygen Demand, or COD. High COD depletes
# Type at least 1 character to search # Hit enter to search or ESC to close

No products in the cart.

No products in the cart.
Product Categories

Oxidation-reduction potential (ORP) is a control parameter frequently used in water treatment systems such as wastewater treatment which works to monitor the effectiveness of the disinfection process. This way, ORP can be used to measure the oxidizing or reducing potential of the wastewater, ensuring the correct amount of additives like chlorine or hydrogen peroxide are added to the wastewater to remove organic and inorganic matter in the water.
Wastewater comes from everyday living in residential and domestic sources, such as our toilets, laundry, bathing, dishwashers etc. After being used the water must be treated and cannot be discharged into land or to surface or groundwater, therefore it goes to wastewater treatment systems.
Wastewater treatment is also important in other industries like pharmaceuticals and food processing to remove impurities, pollutants, and contaminants from the wastewater before it can be discharged into the environment. With a complex process involved, wastewater treatment plants often face many challenges that can impact the efficiency and quality of the cleaning process. Two of the most essential factors that can influence the functioning of wastewater treatment plants are dissolved oxygen (DO) and oxidation-reduction potential (ORP).
Oxidation-reduction potential – ORP (also known as redox), is a vital water quality variable represented by an ORP value that characterizes the redox capacity (electron exchange potential) of the water system.
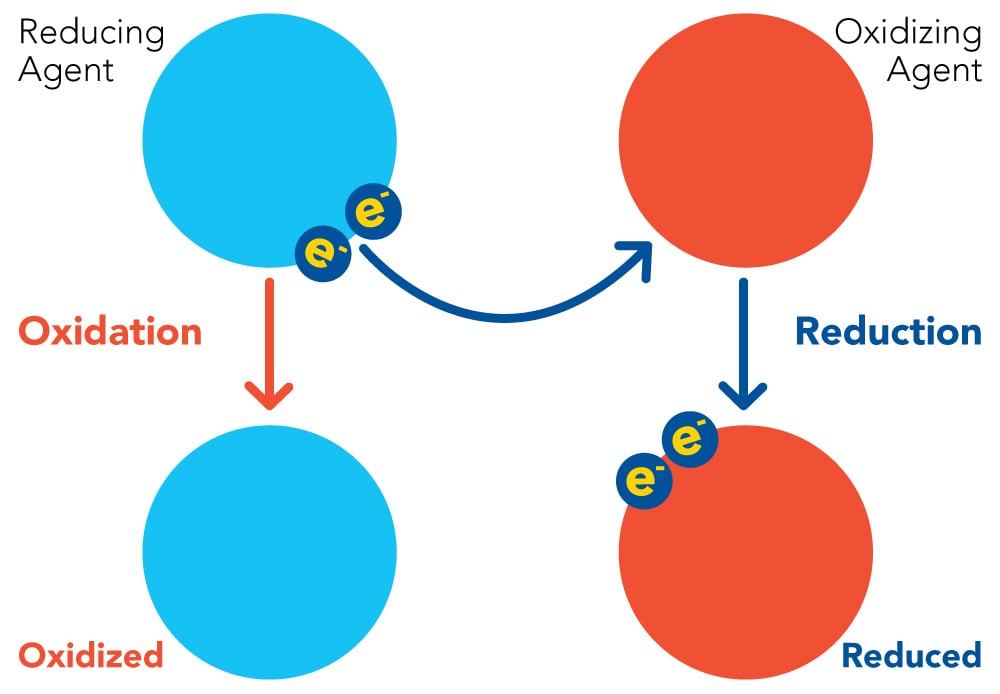
As wastewater treatment processes have many oxidative substances such as dissolved oxygen (DO), nitrite, and nitrate, plus reductive substances like organic matter and ammonium, oxidation-reduction reactions take place in the system. We can work the ORP of the whole wastewater system by measuring the interaction of these reductive and oxidative substances. When the system shows reduction, the ORP value will be higher, and when the ORP value is lower, the ORP value indicates oxidability.
ORP has been used for years to process wastewater generated from metal finishing plants, however, more recently, it has been an essential water quality parameter in municipal wastewater treatment plants.
In wastewater systems, ORP is primarily used to measure reactions that change from oxidized states and reduced states that occur during nitrification, biological phosphorus removal, denitrification, biological malodor production, and the removal of carbon- and hydrogen-compounds, known as cBOD. When an oxidizing agent is present (like oxygen (O2), the ORP value increases, whereas when a reducing agent such as cBOD or a substrate is in the wastewater, the ORP value will decrease. This is a vital piece of information for operators to ensure that the wastewater is treated appropriately, plus it determines what biological reaction is occurring in the wastewater. So, the ORP value tells us the electrochemical activity of the water, which can in turn determine if the wastewater is acidic or alkaline. The ORP scale ranges from -2000 to 2000 mV (negative = reducing conditions and positive = oxidizing conditions).
Let’s put this into an example! So, if a wastewater operator wants to avoid denitrification (clumping) occurring in a secondary clarifier in the treatment plant, the operator needs to maintain an ORP value greater than +50 (millivolts) mV to prevent clumping. On the other hand, if an operator wants to avoid malodor production (wastewater odors like sulfur) in the sewer system of the wastewater treatment plant, the ORP value must be more than -50 mV to prevent sulfide formation. To prevent volatile acid formation an ORP value of more than -100 mV is required.
If inaccurate ORP readings are measured, it can result in chemical imbalances in industrial wastewater treatment processes. For example, if the ORP reading is too high, we know the conditions are oxidizing which can cause the formation of harmful by-products like chloramines which are challenging to remove from wastewater. When low ORP values are measured, the formation of mercaptans and sulfides can occur leading to foul odors and acidic properties in the wastewater.

These chemical imbalances can negatively affect the efficiency and effectiveness of the whole wastewater treatment system. In the long run, these chemical imbalances can also lead to environmental damage, harming humans and the environment.
By accurately measuring ORP, operators can determine the amount of chemical additives needed for the treatment process to return it back into equilibrium. ORP is also essential in reducing the amount of unnecessary chemicals added to the wastewater, minimizing costs for the individual company, and the industry as a whole.

Disinfection is a massive part of industrial wastewater treatments. It guarantees that harmful bacteria and viruses are removed from the wastewater before it is discharged into the environment. By measuring ORP during the disinfection process in wastewater treatment systems, operators can determine the correct amount of disinfectants like chlorine; too much chlorine in water can cause respiratory issues and too little allows harmful bacteria such as salmonella, E. coli, Legionella, and Pseudomonas to remain in the water.
Any industry that discharges wastewater into the environment must follow environmental regulations in the area. ORP measurements play a critical role in ensuring that environmental regulations are complied with and that the wastewater meets acceptable standards before being pumped into the surroundings. In the US, wastewater treatment plants must follow the EPA’s Clean Water Act (CWA). Additionally, wastewater operators must abide by the National Pollutant Discharge Elimination System (NPDES) which issues permits to wastewater treatment facilities.
Various biological processes are involved with wastewater treatment plants to meet regulatory standards and manage bacteria and other pollutants.

Nitrification is an essential process in wastewater treatment systems. The process involves the oxidation of ionized ammonia (NH4+) into nitrate (NO3-). ORP plays an important role in nitrification because the ORP level can influence the activity of nitrifying bacteria. Nitrification in wastewater occurs at ORP levels from +100 to +350 mV.
Denitrification is a critical process in wastewater treatment, essential for meeting total nitrogen discharge limits and controlling filamentous organism growth. It involves denitrifying bacteria reducing nitrate (NO3-) to molecular nitrogen (N2). The oxidation-reduction potential (ORP) is pivotal during denitrification, with optimal activity occurring within an ORP range of the wastewater is +50 to -50 mV.
Biological phosphorus removal is a key process in wastewater treatment plants aimed at meeting total phosphorus discharge limits. It involves two crucial steps: biological phosphorus release and biological phosphorus removal.
In the initial phase of biological phosphorus release, fermentative bacteria generate fatty acids in an anaerobic environment with an ORP ranging from -100 to -225 mV. These fatty acids are then absorbed by phosphorus-accumulating bacteria, prompting the release of phosphorus into the bulk solution.
Subsequently, in the biological phosphorus removal step, phosphorus-accumulating bacteria break down the absorbed fatty acids in an aerobic tank. This breakdown releases energy, which is stored in phosphorus granules. Effective storage necessitates the removal of substantial phosphorus quantities from the bulk solution. This storage process occurs in an aerobic tank with an ORP range of +25 to +250 mV.
Sulfide Formation and Fermentation, also known as Biological Malodor Production, are two significant biochemical processes contributing to unpleasant odors in wastewater treatment. Sulfide formation occurs when sulfate-reducing bacteria degrade sulfate (SO42-), commonly found in groundwater and urine, to produce hydrogen sulfide (H2S). This process, crucially occurring within an ORP range of -50 to -250 mV, is particularly important in anaerobic digesters, where sulfide acts as a nutrient for various anaerobic bacteria, including those responsible for methane production.

Fermentation involves acid-forming bacteria producing volatile acids, nitrogen-containing compounds, and sulfur-containing compounds, many of which contribute to malodors. Fermentation occurs within an ORP range of -100 to -225 mV and plays a vital role in biological phosphorus removal systems, where fatty acid production is necessary for phosphorus release, as well as in anaerobic digesters, where fermentation byproducts are utilized by methane-forming bacteria for methane production.
ORP is critical in both processes as it influences the activity and efficiency of the involved bacteria. Monitoring ORP levels allows operators to maintain optimal conditions for sulfide formation and fermentation, ensuring effective wastewater treatment while mitigating odor issues. Additionally, controlling ORP helps prevent septic conditions that promote sulfide formation, which can lead to the growth of undesirable filamentous organisms, further complicating wastewater treatment processes.
cBOD degradation with free molecular oxygen involves breaking down carbonaceous biochemical oxygen demand (cBOD) using oxygen. ORP is crucial in this process, with optimal degradation occurring within an ORP range of +50 to +250 mV.
Monitoring ORP levels helps regulate conditions for efficient cBOD removal by aerobic bacteria.

Methane production in wastewater involves the generation of methane (CH4) by methane-forming bacteria. It’s favorable in anaerobic digesters but undesirable in sewer systems. Optimal production occurs within an ORP range of -175 to -400 mV.
Understanding the ORP values linked with specific reactions empowers operators to utilize ORP probes effectively in various applications. For example, in sewerage systems, an ORP level below -100 mV indicates malodor production from sulfide formation and fatty acids. Adding sodium nitrate to a manhole elevates the ORP above -50 mV, curbing malodor.
Similarly, monitoring ORP in anaerobic digesters regulates the transfer of nitrate-laden sludge; stopping at -300 mV prevents methane loss as ORP rises from -400 mV. Detecting denitrification absence in tanks via ORP prompts adjustments like altering hydraulic retention time or adjusting cBOD feed to foster denitrification. Monitoring biological phosphorus release in fermentative tanks and extending hydraulic retention time if necessary prevents ORP values exceeding -100 mV.
ORP sensors/probes are essential for monitoring biological reactions in wastewater treatment, aiding operators in identifying acceptable or unacceptable activity. They have become essential tools for wastewater treatment plants, and therefore, operators working in the industry must know how to use them.
As mentioned earlier, to control quality, an ORP device known as a sensor with a probe is inserted directly into the plant’s tank or waste stream to measure electrical charges from ions. The ORP sensor then converts the reading to millivolts (mV) to give a negative or positive reading. Often these ORP meters are used continuously to give operators in the wastewater treatment plant time to identify treatment problems before they could affect effluent quality, ensuring that chemical dosing control is carried out properly.

For reliable ORP measurements, the Atlas Scientific EZO-ORP Sensor Kit is a fundamental tool, offering highly accurate results and interference-free readings.
When using an ORP sensor, calibration is essential to maintain accurate readings. If you are unsure how to calibrate an ORP meter, follow our guide HERE.
In addition to wastewater treatments, ORP is widely used in different applications within industrial water treatments.

Cooling towers are used to recirculate water and remove heat from industrial processes and AC equipment in buildings. These tower systems however are the primary contributors to bacteria growth in water, and therefore frequently monitoring ORP levels is essential. In cooling tower systems, ORP levels greater than 600 mV need to be properly maintained to prevent bacterial growth, in particular, Legionella bacteria which can cause serious pneumonia in humans.
Corrosion can occur in water treatment systems due to many factors such as a lack of dissolved calcium, high concentration of ions (conductivity), a low pH, high temperature, and so on. To prevent corrosion in water systems, operators can maintain ORP measurements to prevent damage to pipes and other water infrastructures.

The agriculture industry in the US accounts for 83.4 million acre-feet of water (NASS, 2018). In agricultural applications, ORP is used to manage the pH level and oxidizing agents used in irrigation. Measuring the ORP level in agricultural irrigation ensures that the water is treated properly to prevent damaging risks to the soil and surrounding environment.
Measuring ORP in wastewater treatment is important as ORP measurements avoid chemical imbalances and ensure that operations follow environmental regulations.

If you want to know more about ORP measurements or the importance of ORP in wastewater treatments, speak to us – the world-class team at Atlas Scientific is more than happy to help!







The amount of oxygen needed to chemically oxidize organic and inorganic materials in wastewater is measured by Chemical Oxygen Demand, or COD. High COD depletes

An RDWC (Recirculating Deep Water Culture) system connects multiple deep water culture sites into one continuously circulating nutrient loop, creating a single, shared root zone.