

Understanding COD in Wastewater Treatment
The amount of oxygen needed to chemically oxidize organic and inorganic materials in wastewater is measured by Chemical Oxygen Demand, or COD. High COD depletes
# Type at least 1 character to search # Hit enter to search or ESC to close

No products in the cart.

No products in the cart.
Product Categories

Water surrounds us constantly in our day-to-day activities. But beyond the normal flow of consuming, bathing, and nature, we often overlook the quality of this liquid life. It drives everything we do from athletics to making a morning cup of joe. We just expect high-quality water, never thinking twice about it. However, from an environmental and manufacturing standpoint, the importance of maintaining water quality cannot be taken as lightly.
So how can you understand good water from bad? Well, the most common water quality measurement to consider is the pH value of water. On one hand, it is a measurement of the hydrogen and hydroxide ion concentration—on the other hand, a straightforward indicator of water quality. So let’s stick with the latter. Running from 0 to 14, the pH scale provides a smaller number to describe acids and a larger number for bases, with 7 being neutral. 7 is lucky for a reason. As a result, high-quality water should hangout around 7 on the pH scale; and since the chemical make-up H2O yields one hydrogen ion (acid) and one hydroxide ion (base) it makes water both an acid and a base (i.e. neutral). However, don’t lose sleep over the exact science because current technology will handle that behind the scenes.

Upkeeping high water quality cannot be understated for the health of everyone from tap water to wastewater dumped into the environment. Speaking of which, the Environmental Protection Agency (EPA) does not regulate pH values directly, but they do recommend a pH range of 6.5 to 8.5 for municipal drinking water (a.k.a tap water for morning joe). Still, by regulating contaminants in water, pH levels are more protected from widely drifting into unsafe levels. For example, acid rain lies around 5 and mild detergent (base) around 10 on the pH scale; since neither can be safely consumed in excess, the small range between them provides motivation to measure and create stable pH levels in water.
We know humans are mostly water, so therefore we require neutral drinking water to avoid unexpected health concerns. So let’s hold our water at lucky number 7 on the pH scale—unless you believe falling into acid will give you superpowers. Although, keep in mind that many manufacturing plants require highly acidic or basic substances to perform an operation (check out how Kevlar is made), which requires in-line monitoring as well as wastewater monitoring of pH. Luckily, pH can be easily measured for proper monitoring thanks to current scientific technology.
Unfortunately, we cannot see ions with the naked eye so counting them is not a feasible option to determine pH levels. One option is the old school litmus papers using qualitative judgments of red and blue to determine whether a solution is acidic or basic. But we need numbers! The next best method would be pH papers that provide a color scale ranging from 0 to 14. Slightly better. However, to create accurate and continuous measurements on a larger scale, pH water testing kits equipped with pH probes will provide a much more robust approach to water’s molecular details.

The most common pH probe, or pH meter, combines two electrodes—a reference and a glass—to measure pH, otherwise known as a combination probe. Since pH is a measure of ions in water, these probes can detect the tiny electrical difference between the probe tips and water.
This is the general concept of most pencil-like laboratory and combination probes. If you are still curious for examples, this concept can be easily observed in many micro pH probes since they have two distinct cords for each electrode. Overall, other measurement approaches occur in different pH sensors for specific applications, but the general concept remains the same: measuring the difference between a reference point and the liquid itself within a given water sample to produce a pH value.

The downside is that this small signal can be disturbed by other factors in the water such as conductivity and other signals from water contamination. Water also has a characteristic called alkalinity, or the ability to resist acidification, which involves other molecules besides H2O. In other words, more pollution requires a more advanced probe to achieve an accurate reading and drown out unwanted signals, or at least a more frequent maintenance schedule for cleaning and calibrating the probes. This issue, more importantly, opens the flood gates to a variety of pH probe styles. The more advanced probes will have higher-grade materials as the probe tip to detect a wider range of pH and to a more accurate degree. Therefore the sensitivity of pH probes all depends on the desired application it will be put to service in—whether it be environmental sampling, household curiosity, continuous pipe flow measurement, etc.
As discussed above, pH water test kits will mainly consist of a probe, calibration solution, and storage solution. If the probe itself does not have a digital readout then the kit should supply equipment to convert the probe reading to a pH value on common digital monitors like your computer or even a television screen. This more customizable pH probe setup is generally used for industrial and commercial laboratory applications when continuous and detailed readout is required. For example, a water treatment plant could mount a pH probe directly into a pipe for continuous readout, or a laboratory could be ad hoc measuring changes in pH from different chemical reactions. Again, it is very important to maintain moisture for any application so that the probe doesn’t dry and become useless. For industrial applications and pipe mounting purposes, extra considerations will need to be considered to prevent probe dry out. That said, water pH test kits are the best option for determining water pH for many applications from a personal pool all the way up to industrial facilities. Some common pH probes offered at Atlas Scientific include:
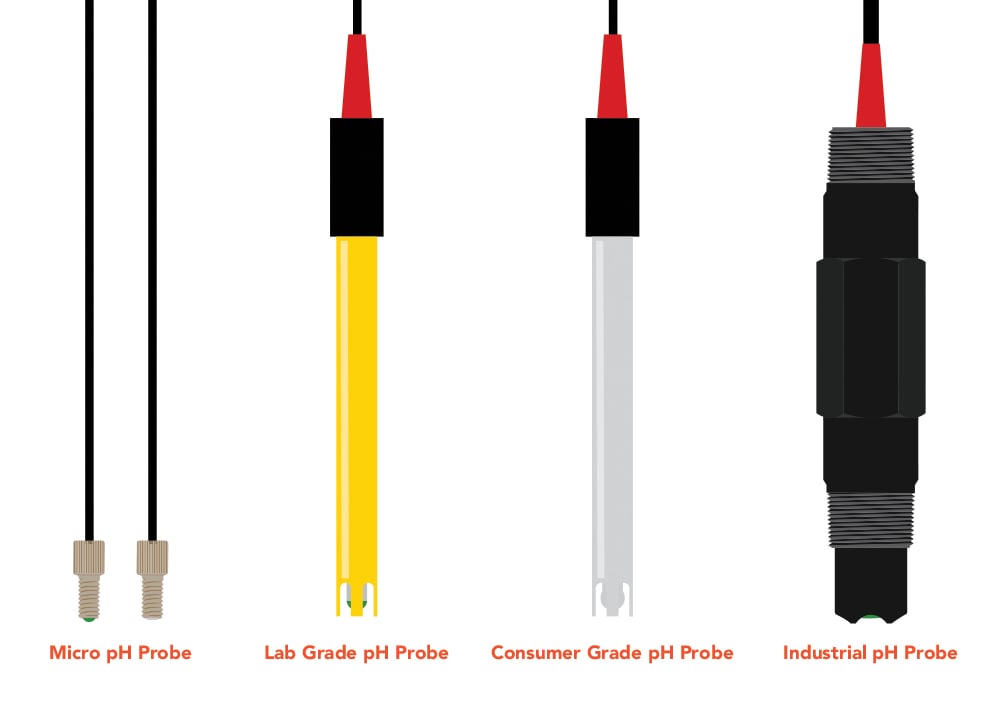
These regular pH probes come in different grades from consumer to lab (research) grade, depending on the level of accuracy required and environment expected. It is important to understand the pH application before choosing a probe to ensure the equipment will meet your needs. That said, there’s no reason to use a research-grade probe on the backyard pool unless it’s a quick afternoon borrow from Carl, the scientist, across the street. The higher-grade probe will work, of course, but the lab grade equipment is more applicable in larger ranges of pH—where substances become highly acidic or basic. In this pool test scenario, a consumer-grade pH probe will work wonders since pools hangout around 7-8 on the pH scale, but it could not handle a highly acidic titration experiment (think Breaking Bad chemistry-esque). Simply stick the probe into the liquid and a pH value should display on a readout screen quicker than you can say “Carl, the scientist.” Note that the pH probe itself does not provide a pH value, but the combination of the probe and EZO-pH circuit outputs a pH reading onto a screen. Either way, both probe grades are easily calibrated and connected to a readout screen by purchasing a water pH test kit.
Since micro pH probes are so small they are conducive to research applications in microfluidics and other small volume applications where large pH probes would be pointless. In other words, a sledgehammer would be wasted on placing a thumbtack into a corkboard. These probes are so small they require two separate cords for each electrode, as discussed earlier, which streamline into one output for a pH reading. For everyday use from pool monitoring to environmental sampling the regular pH probe will suffice in that it provides accurate, low-interference signal and extreme ease of use.
The industrial probe will be a step up from the regular pH probe to increase usability conditions with its highly durable probe housing and advanced sensing materials. In other words, this type of probe can handle all the harsh conditions of a wastewater plant and other intense industrial applications. The sledgehammer of pH probes. It additionally offers the ability to screw directly into larger piping systems for continuous monitoring. Specifically, the industrial probe at Atlas Scientific has a ¾ inch national pipe thread (NPT). This industrial probe grade also has an overachieving cousin that measures temperature and oxidation-reduction potential (ORP) on top of the pH measurements. In any scenario, the industrial pH will perform and keep performing in any situation thrown its way.
Keeping close tabs on water quality and pH readings will aid any application (consumer, industrial, laboratory) to reduce waste and downtime, save energy, maintain a healthy environment, and achieve sustainability regulations. With pollution becoming an ever-increasing issue, the quick measurement of pH provides instant feedback into further investigation of contaminants that would harm human and environmental health.
Even further, quality water will reduce the risk of human health concerns like dry skin and hair, itching, and irritation—problems nobody wants to deal with retroactively. The key will be to maintain a neutral pH for most water processes, but if highly acidic or basic conditions are required then effluent water must be highly monitored using none other than a pH probe as discussed above. In addition, the use of multiple probes will never hurt your effort to achieve quality water.
Again, one must consider the intended use before deciding on the correct pH probe. In one scenario, consumers could purchase the lowest grade regular pH probe to test a swimming pool or tap water to gain insight on pH. These tests will offer knowledge about whether or not their water will be hard and/or corrosive to piping systems and human health. In a similar manner, this corrosion consideration also applies to industrial applications but these buyers should consider the industrial probes to have continuous monitoring of pH for more intense ranges of pH. Proactive monitoring will ensure proper signals to maintain quality water before pH sways too far in either direction because large corrections to pH can accrue a large chemical cost. In reality, it’s much better to invest in a water pH test kit than to cut into the rainy day fund.
To summarize, you will need to first identify the pH measurement requirements specific to your application. Environmental samples? Continuous monitoring? Backyard pool party? In almost every instance you will require a water pH test kit to achieve accurate readouts and monitoring. By identifying the desired durability and accuracy specifications, it will be much easier to make a strong and informed decision when choosing a pH probe and water pH test kit that will save you money in the future.
If you are unsure exactly which water pH test kit will best suit your needs, do not hesitate to reach out to the world-class team at Atlas Scientific.







The amount of oxygen needed to chemically oxidize organic and inorganic materials in wastewater is measured by Chemical Oxygen Demand, or COD. High COD depletes

An RDWC (Recirculating Deep Water Culture) system connects multiple deep water culture sites into one continuously circulating nutrient loop, creating a single, shared root zone.